Products
홈
Products
Himlaca®
Himlaca®

Products 01
 Scientifically proven Centella Asiatica extract for Eye, Liver, and Cognitive health
Scientifically proven Centella Asiatica extract for Eye, Liver, and Cognitive health
Functional Health Claim
- Support eye health by maintaining the density of macular pigments which can be affected by aging
- Support liver health by preventing non-alcoholic liver injury
- Support cognitive health by preventing cognitive decline and memory impairment
Dosage
- 300mg/day as Centella Asiatica extract
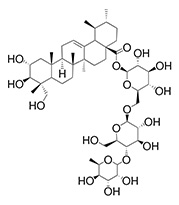
Active Compound
| Name | Asiaticoside |
|---|---|
| Molecular Formula | C48H78O19 |
| Molecular Weight | 959.1 |
| CAS No. | 16830-15-2 |
Clinical Trial
1Eye Health
- Subject80 elderly male or female participants aged 45-65 years, with low MPOD levels (0.2 to 0.4)
- MethodTake Himalca®(Centella Asiatica extract) 300 mg/day for 180 days
A double-blind, randomized, and placebo-controlled study
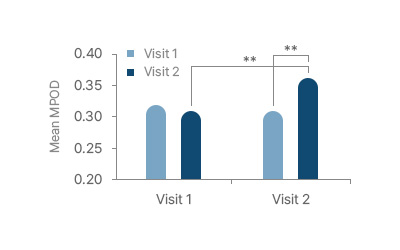
MPOD improvement beginning after 60 days (visit 2)
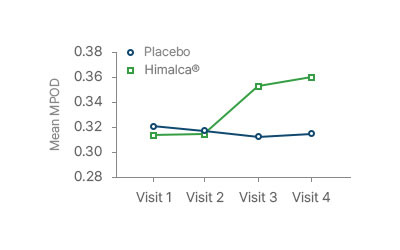
MPOD was significantly improved after 180 days
compared to the control group (P < 0.01)
Nutrients 2026, 18, 905
2Liver Health
- Subject80 elderly male or female participants aged 20-70 years, with ALT level 45 ~ 135 U/L and AST level 25 U/L or higher in the previous 2 weeks
- MethodTake Himalca®(Centella Asiatica extract) 300 mg/day for 180 days
A Randomized, Double-Blind, Placebo-Controlled Parallel study
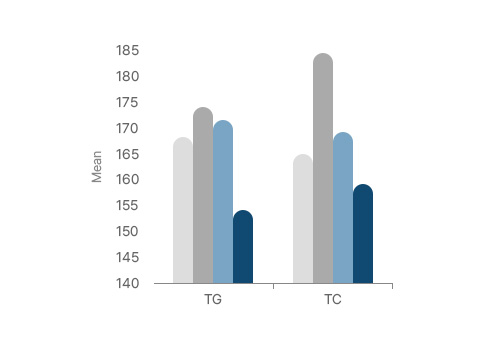
- Before (Placebo)
- After (Placebo)
- Before (Himalca®)
- After (Himalca®)
ALT decreased 36.7%
AST decreased 27%
GGT decreased 18.8%
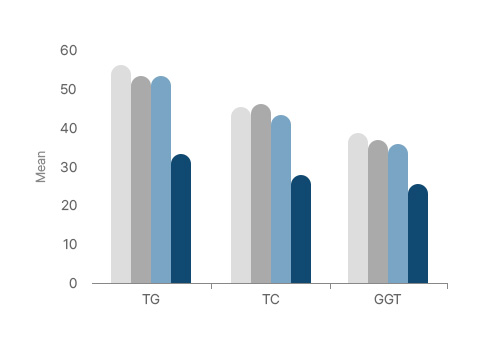
Lipid metabolism-related markers
TG and TC significantly decreased
Appl. Sci. 2021, 11, 11498.
3Cognitive health
- - TBU -
Patents
| Patent name | Registration status |
|---|---|
| Composition for preventing or treating retinal disease comprising compounds isolated from Centella asiatica extract | KP Registered |
-

KP10-2284682
(2021.07.27)
| Patent name | Registration status |
|---|---|
| Composition for preventing or treating retinal disease, containing Centella asatica extract | KP, EP, JP Registered PCT, US, CN Pending |
-

KP10 -2181864
(2020.11.17) -

EP3868218
(2023.05.07) -

JP07648529
(2025.03.10)